|
#1
| |||
| |||
|
hello sir, i am in final yr b.pharm . i searched over intenet but could not get the soft copy of final yr syllabus for B.Pharm . kindly if u can send me the same it would be very helpful n appreciated.as my exams are on the the run, if i could get it as soon as posible would be very helpful. Thank you. Regards Gibran gibran.sonu@gmail.com gibran_sonu@yahoo.in As you want to get syllabus for B.Pharma Final Year of University of Pune, so here I am providing the following syllabus: University of Pune B.Pharma Final Year Syllabus 4.1 (T) PHARMACEUTICS-III SECTION-I Sterile formulations i) Preformulation Methodology for Parenteral Products : Physicochemical properties of drug substances, accelerated stability study, preformulation studies for proteins, peptides ii) General Requirements : Routes of administration, significance of tonicity adjustment and sterility, freedom from pyrogens and particulate matter, stability aspects and quality control tests, sterility tests for ointment, antibiotic preparations, powders. iii) Small Volume Parenterals (SVPs) : Classification, formulation of solutions and suspensions, types of vehicles, selection of vehicles and added substance, processing and manufacturing of SVPs, Pilot plant scale up for SVPs. Special types of SVPs: Formulation of peptides and proteins, freeze dried products, parenteral suspensions, emulsions and Reconstituted products. iv) Large Volume Parenterals (LVPs): Types of LVPs, concept of formulation, influence of physiological, formulation and packaging parameters, stabilization of LVPs, processing of LVPs, Total Parenteral Nutrition (TPN) and Peritoneal dialysis fluid. Pilot plant scale up for LVPs. v) Ophthalmic Products : General requirements, formulation, types of dosage forms. Contact lens and lens care products. vi) Packaging of Parenterals : Various materials used, official quality control tests, packaging components and types, specifications and methods of evaluation, stability, factors influencing choice of containers, prefilled syringes, blow-fill-seal technique. vii) GMP-Design of Parenteral Production Facility: Product characteristics, water treatment plant, operational assessment and area planning, batch Vs continuous operation, environmental control zones, filling area design, utility distribution systems, heating ventilation air conditioning (HVAC), HEPA filter testing and rating, laminar flow area working, development of facility layout, automation in parenteral industry. University of Pune B.Pharma Final Year Syllabus     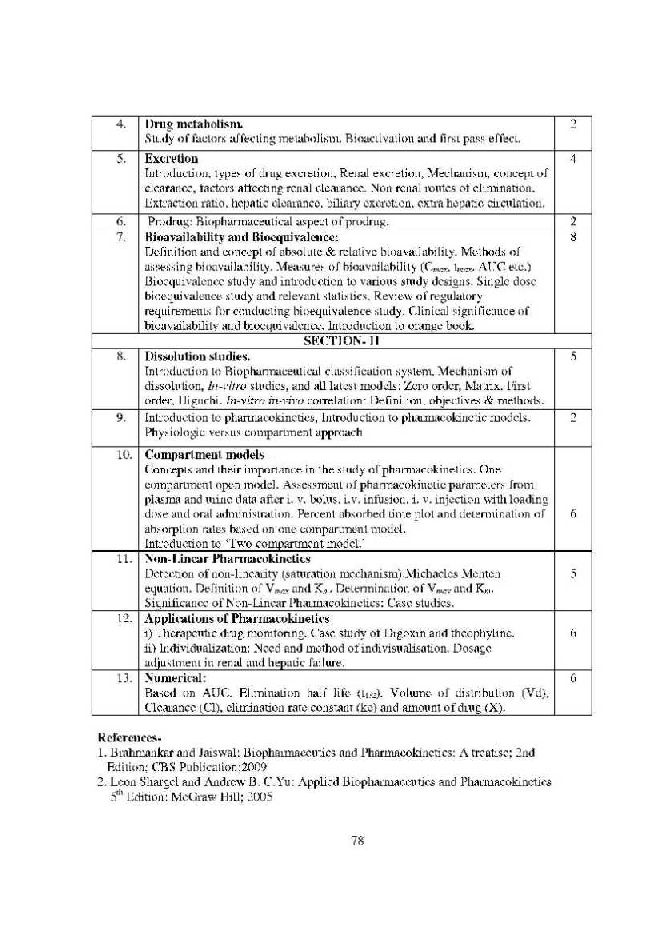     Contact Details University Of Pune Ganeshkhind, Pune, Maharashtra 411007 Map  Last edited by Aakashd; October 16th, 2019 at 04:43 PM. |