|
#1
| |||
| |||
|
Can you give me the complete details of NDA New Drug Application and tell me from where I can download it. Give me more information about it and its uses |
|
#2
| |||
| |||
|
I am providing the information about the NDA New Drug Application for your reference. You can go through it. I am providing the information in a power point presentation for you. You can use it for preparations.
__________________ Answered By StudyChaCha Member |
|
#4
| |||
| |||
|
The Food and Drug Administration's New Drug Application is the vehicle in the United States through which drug sponsors formally propose that the FDA approve a new pharmaceutical for sale and marketing. A new drug application (NDA) is a comprehensive document that must be submitted to the U.S. Food and Drug Administration (FDA) in order to request approval for marketing a new drug in the United States. Please find the below attached file for details about NDA New Drug Application; NDA drug approval process 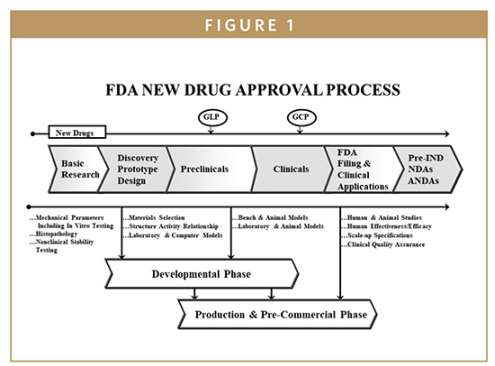 The FDA filing and premarket applications consist of the following categories: 1. Investigational New Drug Application (IND) 2. New Drug Application (NDA) 3. Abbreviated New Drug Application (ANDA)
__________________ Answered By StudyChaCha Member |
 |
| |