|
#14
| |||
| |||
|
As you need the B.Sc. Chemistry UG course syllabus of the Thiruvalluvar University so here I am providing the same Objective: Basic concepts regarding atomic structure, periodic properties, bonding concepts, ionic bond, VSEPR and MO theories, nomenclature of organic compounds, hybridization, reaction intermediates, quantum theory, gases, principles of volumetric analysis, related problems, and applications wherever necessary are to be taught for I-Semester. UNIT-I 1.1 Atomic structure - Quantum numbers n, l, m and s - Pauli exclusion principle – Energy distribution and orbitals - Hund’s rule of maximum multiplicity - Aufbau's principle – Electronic configurations of elements - Stability of half-filled and completely filled orbitals. 1.2 Classification of elements – General characteristics of s, p, d and f block elements -Periodicity of properties- Definition and periodicity of the following properties- Atomic radii - factors affecting atomic radii - Ionic radii - factors affecting ionic radii. 1.3 Ionization potential - factors affecting ionization potential - Electron affinity - factors affecting electron affinity - Electronegativity - factors affecting electronegativity - Pauling scale – Mulliken electronegativity scale – Applications of electronegativity regarding bonding nature. UNIT-II 2.1 Ionic bond - Conditions for the formation of ionic bond - General properties - Energetics of formation of NaCl from Na+ and Cl -. Hydration energy and lattice energy and their applications – Born – Haber cycle. Fajan's rules - Characteristics of electrovalent compounds - Valence bond Thiruvalluvar University B.Sc. Chemistry Syllabus 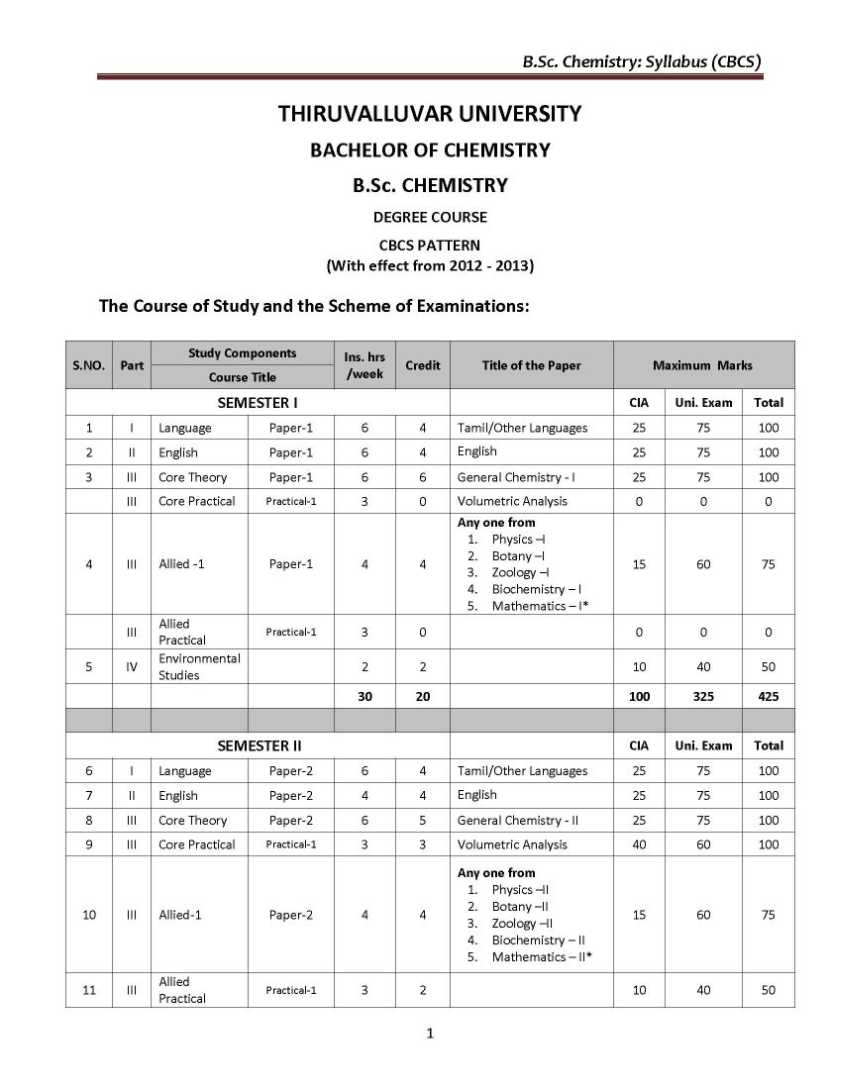 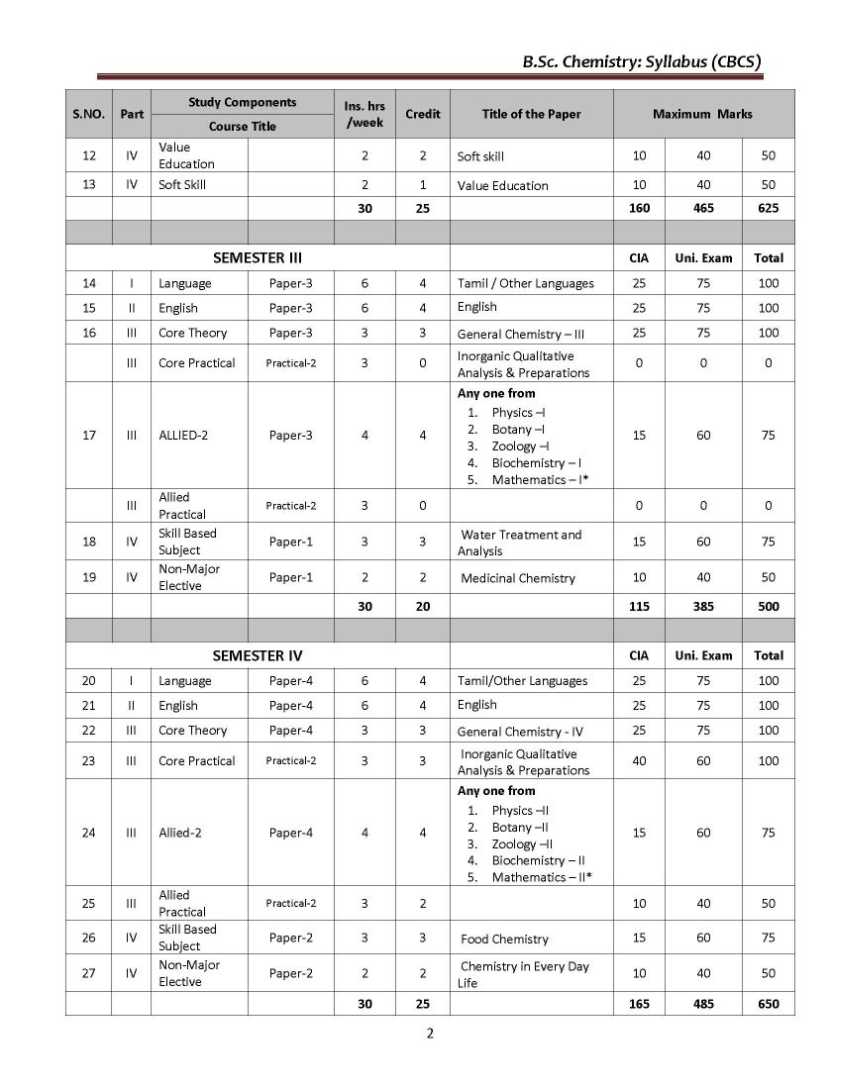 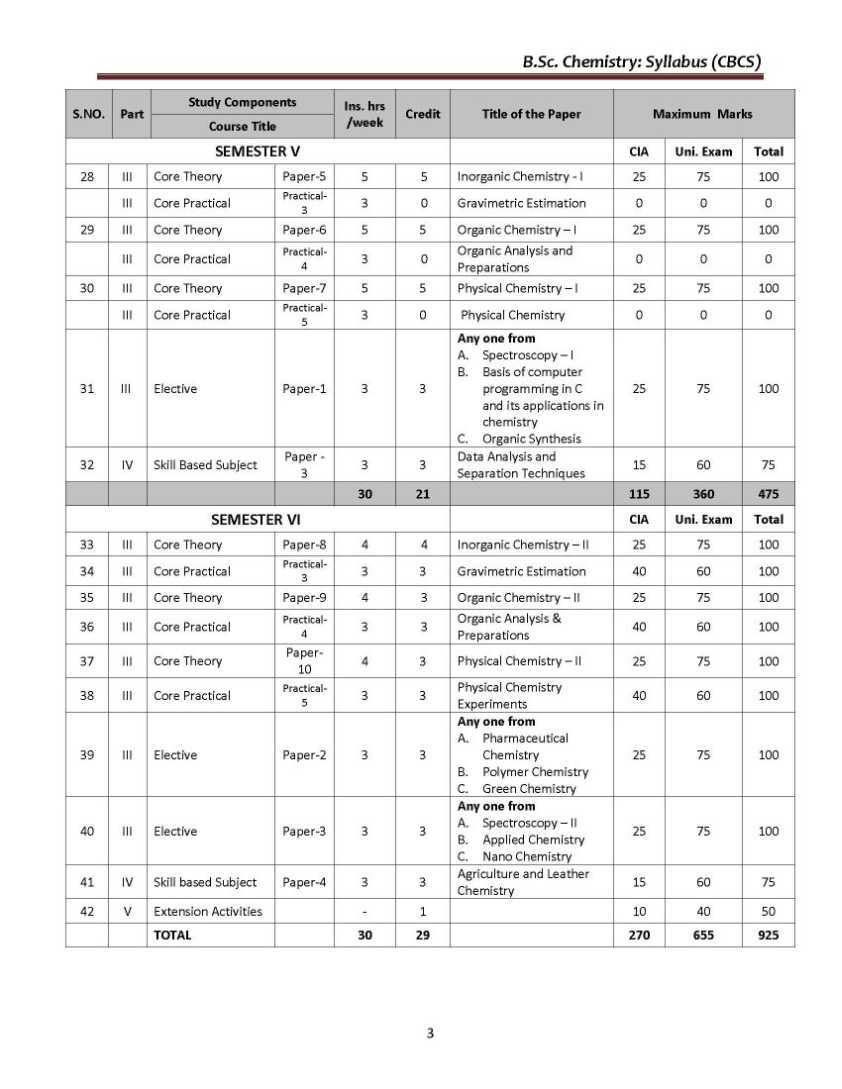 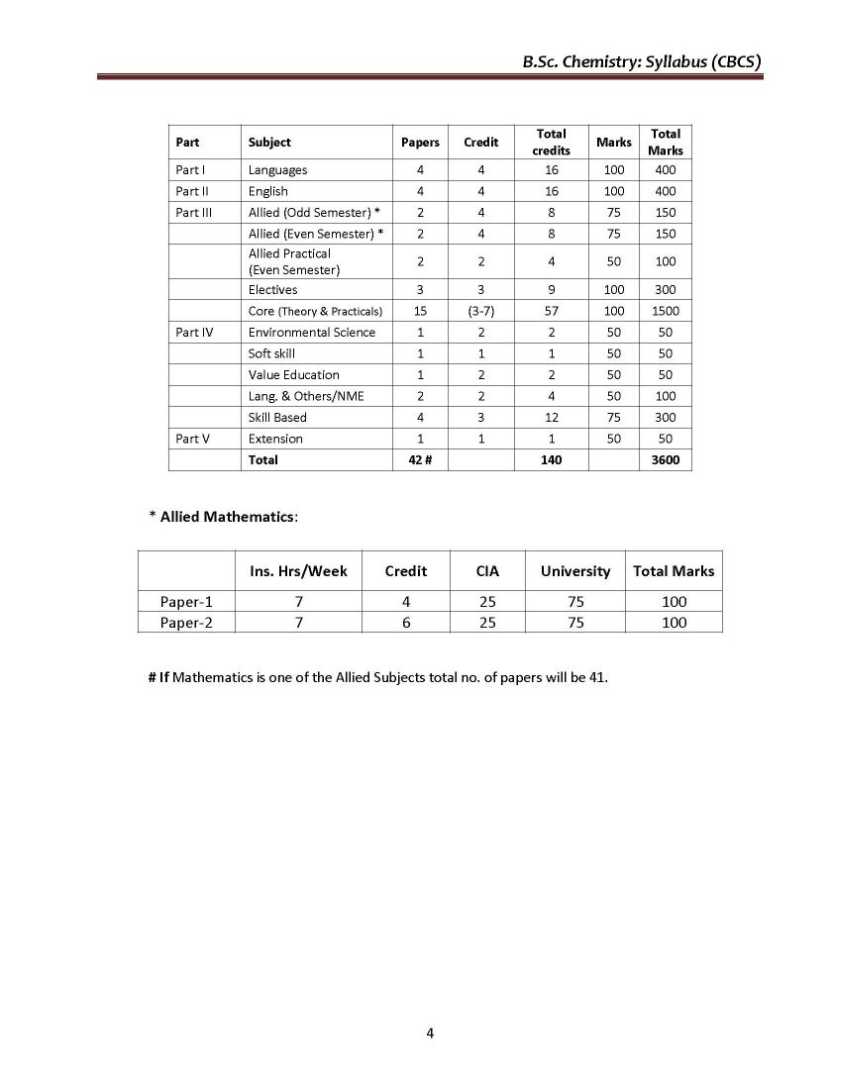  For the complete syllabus here is the attachment;
__________________ Answered By StudyChaCha Member |