|
#1
| |||
| |||
|
Hi I would like to have the information about the Vibration- Rotation Spectroscopy of HCl and DCl as well as the details about its theory and procedure and also Isotope Effects?
|
|
#2
| |||
| |||
|
Vibration spectroscopy is a standout amongst the most essential instruments for the exact assurance of sub-atomic structure. Vibration spectroscopy additionally assumes an imperative part in ecological science. For instance, the albedo of the world's climate is controlled by the assimilation of infra-red light by green house gasses, which incorporate CO2, H2O, and CH4. For typical modes that have a changing dipole minute, the vibrational range of an atom is detectable utilizing infrared assimilation. Such modes are said to be IR dynamic. Raman spectroscopy is utilized to watch typical modes that are not IR dynamic. The principal vibration recurrence of a typical mode is subject to the isotopes of the particles that are included. In this way, vibration spectroscopy is a extremely valuable approach to consider isotope impacts. The move in essential vibration recurrence and rotational steady with isotopic substitution will likewise be anticipated and contrasted with the exploratory esteems. Reason: To decide the basic vibration recurrence and bond length for H35Cl, H37Cl, D35Cl, and D37Cl and to contrast the isotope impacts with hypothetically anticipated esteems. Isotope Effects: H35Cl, H37Cl, D35Cl, and D37Cl each have exceptional spectra on the grounds that the substitution of various isotopes changes the lessened mass of the particle, Eq. 4. H particles have the biggest change in isotopic mass of all iotas, a factor of ~2 in substituting D (i.e. 2H) for H (i.e. 1H). The investigation of isotope impacts, particularly upon deuteration is a vital device in unthinking natural science. From Eq. 3, hypothesis predicts that the proportion of the key vibration recurrence for two unique isotopically substituted adaptations of the same molecule,1 and 2 Theory & Procedure Details  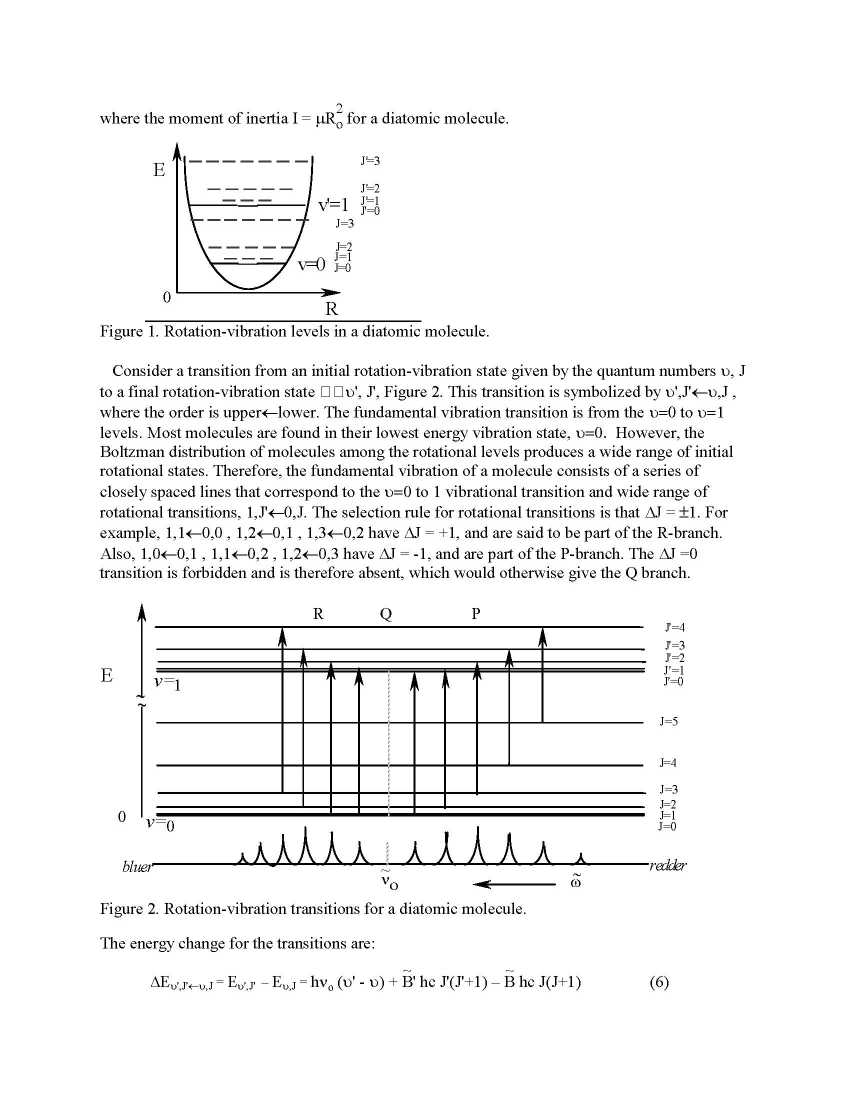 
__________________ Answered By StudyChaCha Member |