|
#1
| |||
| |||
|
I am searching for CBSE class 12th Biology course syllabus so how can get it?
|
|
#2
| |||
| |||
|
You want CBSE class 12th Biology course syllabus so I have and I am sharing it with you: II. Genetics and Evolution (45 Periods) Heredity and variation: Mendelian Inheritance; Deviations from Mendelism-Incomplete dominance, Co-dominance, Multiple alleles and Inheritance of blood groups, Pleiotropy; Elementary idea of polygenic inheritance; Chromosome theory of inheritance; Chromosomes and genes; Sex determination - in humans, birds, honey bee; Linkage and crossing over; Sex linked inheritance - Haemophilia, Colour blindness; Mendelian disorder in humans - Thalassemia; chromosomal disorders in humans; Down's syndrome, Turner's and Klinefelter's syndromes. Molecular Basis of Inheritance: Search for genetic material and DNA as genetic material; Structure of DNA and RNA; DNA packaging; DNA replication; Central dogma; Transcription, genetic code, translation; Gene expression and regulation - Lac Operon; Genome and human ganeome project; DNA finger printing. Evolution: Origin of life; Biological evolution and evidences for biological evolution (Paleontological, comparative anatomy, embryology and molecular evidence); Darwin's contribution, Modern Synthetic theory of Evolution; Mechanism of evolution – Variation (Mutation and Recombination) and Natural Selection with examples, types of natural selection; Gene flow and genetic drift; Hardy - Weinberg's principle; Adaptive Radiation; Human evolution. Rest of syllabus is in attached pdf file which you can free download. I also have class 12th following subjects syllabus which I can share with you if you want: Hindi Aichik Hindi Part: B Hindi Core English Elective English Core English Functional Mathematics Biology Physics Chemistry New!Accountancy New!History Geography Business Studies Enterprenuership Economics |
|
#4
| |||
| |||
|
As you want to get CBSE Board 12th chemistry Syllabus so here I am giving you same: Unit I: Solid State 10 Periods Classification of solids based on different binding forces: molecular, ionic, covalent and metallic solids, amorphous and crystalline solids (elementary idea). Unit cell in two dimensional and three dimensional lattices, calculation of density of unit cell, packing in solids, packing efficiency, voids, number of atoms per unit cell in a cubic unit cell, point defects, electrical and magnetic properties. Band theory of metals, conductors, semiconductors and insulators and n and p type semiconductors. Unit II: Solutions 10 Periods Types of solutions, expression of concentration of solutions of solids in liquids, solubility of gases in liquids, solid solutions, colligative properties - relative lowering of vapour pressure, Raoult's law, elevation of boiling point, depression of freezing point, osmotic pressure, determination of molecular masses using colligative properties, abnormal molecular mass, Van't Hoff factor. Unit III: Electrochemistry 12 Periods Redox reactions, conductance in electrolytic solutions, specific and molar conductivity, variations of conductivity with concentration, Kohlrausch's Law, electrolysis and law of electrolysis (elementary idea), dry cell-electrolytic cells and Galvanic cells, lead accumulator, EMF of a cell, standard electrode potential, Nernst equation and its application to chemical cells, Relation between Gibbs energy change and EMF of a cell, fuel cells, corrosion. Unit IV: Chemical Kinetics 10 Periods Rate of a reaction (Average and instantaneous), factors affecting rate of reaction: concentration, temperature, catalyst; order and molecularity of a reaction, rate law and specific rate constant,integrated rate equations and half life (only for zero and first order reactions), concept of collision theory (elementary idea, no mathematical treatment). Activation energy, Arrhenious equation. Unit V: Surface Chemistry 08 Periods Adsorption - physisorption and chemisorption, factors affecting adsorption of gases on solids, catalysis, homogenous and heterogenous activity and selectivity; enzyme catalysis colloidal state distinction between true solutions, colloids and suspension; lyophilic, lyophobic multimolecularand macromolecular colloids; properties of colloids; Tyndall effect, Brownian movement,electrophoresis, coagulation, emulsion - types of emulsions. CBSE Board 12th chemistry Syllabus 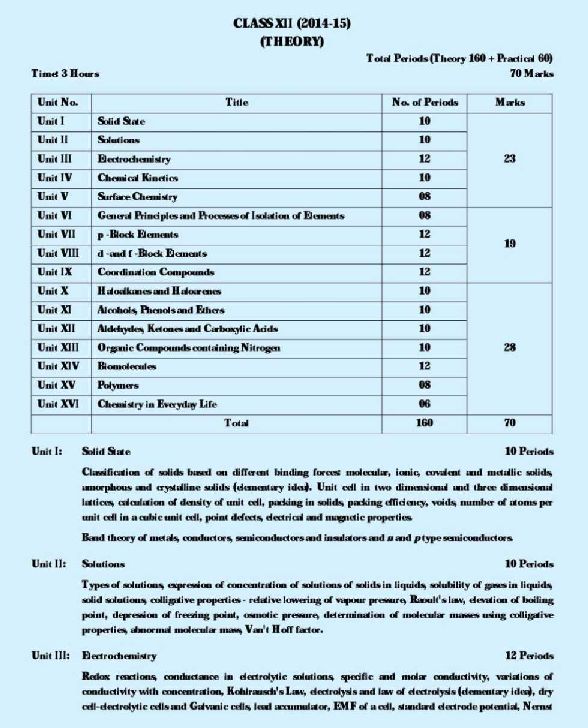     For full syllabus here is the attachment.........................
__________________ Answered By StudyChaCha Member Last edited by Aakashd; August 4th, 2018 at 07:54 AM. |